1 in 6 ED visits resulted in a prescription for buprenorphine, extended-release naltrexone, or methadone within 30 days.
Research finds that medications for opioid use disorder were rarely prescribed following emergency department visits for opioid overdose among patients with Medicaid. In this IHPI brief, a U-M team outlines considerations for policymakers and health systems to improve treatment initiation rates.
A record 81,000 Americans died from opioid overdose in 2021.1
Many of these deaths could be prevented with medications for opioid use disorder, including buprenorphine, methadone, and extended-release naltrexone — three FDA-approved medications to treat opioid use disorder (OUD).2–4
Patients with OUD account for 1 in 80 emergency department (ED) visits each year.5,6 These visits represent valuable opportunities to initiate medications for OUD or to encourage follow-up with a clinician who can prescribe these medications.7
Prior studies, however, have found that treatment initiation remains limited following ED visits for opioid overdose. For example, one study found buprenorphine was prescribed within 30 days after just one out of 12 ED visits.8 A variety of barriers contribute to low rates of treatment initiation, including an inadequate number of professionals offering treatment in the community, gaps in OUD medication training and resources for health providers, and ineffective referral systems for addiction care between EDs and outpatient treatment facilities.
Medicaid plays a key role in facilitating access to behavioral health services for some of the most vulnerable populations in the U.S., including mental health and substance use disorder treatment, as the program provides health coverage to 38% of adults under 65 years old with opioid use disorder.9,10
But there is limited national data on the rates of initiation of medications for opioid use disorder following an ED visit for opioid overdose in the Medicaid population. To address this gap in knowledge, a research team from the University of Michigan studied dispensing of medications for OUD among beneficiaries covered by Medicaid, looking at variation by geographic region and state.
Takeaways from our study
Our team studied initiation of medications for opioid use disorder following an ED visit for opioid overdose using nationwide Medicaid claims data. The dataset included nearly 72,000 Medicaid beneficiaries aged 12–64 years who had an ED visit for opioid overdose in 2018. Treatment initiation was defined as receiving a prescription by any provider for buprenorphine, extended-release naltrexone, or methadone for OUD within 30 days of discharge.
Medications for opioid use disorder were rarely prescribed following ED visits for opioid overdose among patients with Medicaid coverage.
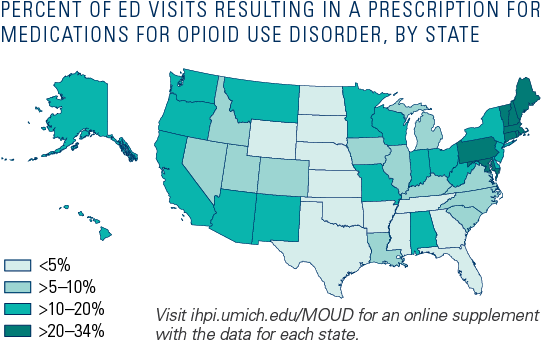
Rates of treatment initiation following ED visits for opioid overdose varied widely by region and state.
PERCENT OF ED VISITS RESULTING IN A PRESCRIPTION FOR MEDICATIONS FOR OPIOID USE DISORDER, BY REGION
Nationwide: 17%
Northeast: 24%
Midwest: 13%
South: 15%
West: 15%
What are the implications for policy and practice?
Our study shows that rates of initiation of medications for OUD following ED visits for opioid overdose are low across all regions in the U.S. in the Medicaid population but are particularly low in the Midwest, South, and West. The findings also show that better performance is possible, as eight states—seven of which are in the Northeast—had initiation rates between 20–34%, compared to the national average of 17%. Identifying why these states are performing better could inform strategies to improve initiation rates after ED visits for opioid overdose.
While there are many efforts underway to address low treatment initiation rates, further research and evaluation is needed to better understand the effect on initiation rates and share best practices and lessons learned across states and communities.
The U-M research team outlined several considerations to help improve treatment initiation rates:
Policymakers: As geographic barriers and health workforce shortages contribute to a lack of adequate outpatient substance use disorder treatment options in many communities, states could explore various strategies to enhance treatment options for patients with opioid use disorder. They could consider improving access to addiction care through allowing telemedicine visits11, facilitating linkages between health systems and community-based services to support the continuum of care, expanding the use of non-physician providers to prescribe treatment12, or taking steps to increase the overall number of professionals offering treatment in the community. Of note, initiation and engagement of substance use disorder treatment are among the behavioral health measures which will become mandatory to be reported for Medicaid by states in 2024.13 Policymakers could evaluate and adapt those measures to further improve healthcare processes and patient outcomes going forward.
Additionally, policymakers could provide resources and incentives for hospitals to encourage treatment initiation models of care in the ED and outpatient settings, such as training or technical assistance. For example, CA Bridge is a statewide program in California that provides hospitals with funding for clinician champions, substance use navigators, and technical assistance to support implementation, clinician training, and treatment protocols in the ED.14
To address patient barriers to treatment initiation, state Medicaid agencies and legislatures could consider adjusting prior authorization requirements for clinicians to prescribe any of these medications, including extended-release buprenorphine. For example, a study by a U-M research team found that, in 2017–2021, 37% of Medicaid formularies imposed prior authorization requirements for extended-release buprenorphine, compared to 5% for immediate-release buprenorphine.15 The use of extended-release buprenorphine, a monthly dosing medication, could help address treatment adherence barriers that many patients experience.
Hospitals and health systems: Hospitals and health systems could implement streamlined treatment initiation protocols after an opioid overdose to improve initiation rates using a multifaceted approach. They could invest in training programs for ED and outpatient clinicians, including primary care providers and addiction specialists, to help improve their knowledge of local treatment options and resources. They could also enhance linkages between EDs and outpatient settings to connect patients to care following discharge. The CA Bridge program is an example of this approach and has worked effectively to expand medication for addiction treatment. Future research is needed to comprehensively evaluate the impact of such programs on medication initiation rates and patient outcomes.
|
State |
State abbreviation |
Region |
Number of ED visits for opioid overdose |
Number of ED visits that resulted in initiation of medications for OUD within 30 days |
Percent of visits that resulted in initiation |
|
Rhode Island |
RI |
Northeast |
301 |
103 |
34.2 |
|
Massachusetts |
MA |
Northeast |
5,693 |
1,835 |
32.2 |
|
Maryland |
MD |
South |
4,623 |
1,481 |
32 |
|
Vermont |
VT |
Northeast |
229 |
69 |
30.1 |
|
Maine |
ME |
Northeast |
178 |
52 |
29.2 |
|
Connecticut |
CT |
Northeast |
2,789 |
724 |
26 |
|
Pennsylvania |
PA |
Northeast |
4,810 |
1,076 |
22.4 |
|
New Hampshire |
NH |
Northeast |
335 |
70 |
20.9 |
|
Delaware |
DE |
South |
1,110 |
221 |
19.9 |
|
Washington |
WA |
West |
2,818 |
553 |
19.6 |
|
Oregon |
OR |
West |
969 |
185 |
19.1 |
|
Ohio |
OH |
Midwest |
8,197 |
1,537 |
18.8 |
|
New Mexico |
NM |
West |
966 |
180 |
18.6 |
|
Minnesota |
MN |
Midwest |
1,148 |
212 |
18.5 |
|
Hawaii |
HI |
West |
111 |
20 |
18 |
|
Wisconsin |
WI |
Midwest |
962 |
165 |
17.2 |
|
New Jersey |
NJ |
Northeast |
2,069 |
351 |
17 |
|
New York |
NY |
Northeast |
4,606 |
773 |
16.8 |
|
California |
CA |
West |
5,637 |
851 |
15.1 |
|
Indiana |
IN |
Midwest |
1,766 |
261 |
14.8 |
|
Missouri |
MO |
Midwest |
687 |
96 |
14 |
|
District of Columbia |
DC |
South |
330 |
46 |
13.9 |
|
Alabama |
AL |
South |
185 |
23 |
12.4 |
|
Alaska |
AK |
West |
175 |
20 |
11.4 |
|
Arizona |
AZ |
West |
1,850 |
208 |
11.2 |
|
Montana |
MT |
West |
150 |
16 |
10.7 |
|
West Virginia |
WV |
South |
775 |
82 |
10.6 |
|
Kentucky |
KY |
South |
3,609 |
335 |
9.3 |
|
Iowa |
IA |
Midwest |
329 |
29 |
8.8 |
|
Utah |
UT |
West |
137 |
12 |
8.8 |
|
Virginia |
VA |
South |
469 |
41 |
8.7 |
|
Colorado |
CO |
West |
1,352 |
118 |
8.7 |
|
Michigan |
MI |
Midwest |
5,155 |
421 |
8.2 |
|
Illinois |
IL |
Midwest |
4,384 |
345 |
7.9 |
|
South Carolina |
SC |
South |
425 |
32 |
7.5 |
|
Louisiana |
LA |
South |
2,029 |
145 |
7.1 |
|
Idaho |
ID |
West |
71 |
5 |
7 |
|
North Carolina |
NC |
South |
980 |
66 |
6.7 |
|
Nevada |
NV |
West |
590 |
33 |
5.6 |
|
Texas |
TX |
South |
352 |
17 |
4.8 |
|
Mississippi |
MS |
South |
95 |
4 |
4.2 |
|
Kansas |
KS |
Midwest |
125 |
5 |
4 |
|
Georgia |
GA |
South |
289 |
11 |
3.8 |
|
Tennessee |
TN |
South |
1,061 |
40 |
3.8 |
|
Oklahoma |
OK |
South |
166 |
6 |
3.6 |
|
North Dakota |
ND |
Midwest |
61 |
2 |
3.3 |
|
Florida |
FL |
South |
162 |
3 |
1.9 |
|
Arkansas |
AR |
South |
162 |
2 |
1.2 |
|
South Dakota |
SD |
Midwest |
21 |
0 |
0 |
|
Wyoming |
WY |
West |
18 |
0 |
0 |
|
Nebraska |
NE |
Midwest |
48 |
0 |
0 |
1. Provisional Drug Overdose Death Counts. Ahmad F, Rossen L, Sutton P. National Vital Statistics System. Accessed May 9, 2022. https://www.cdc.gov/nchs/nvss/vsrr/drug-overdose-data.htm.
2. Mortality Among Clients of a State-Wide Opioid Pharmacotherapy Program Over 20 years: Risk Factors and Lives Saved. Degenhardt L, Randall D, Hall W, et al. Drug Alcohol Depend. 2009;105(1-2):9–15. doi:10.1016/j.drugalcdep.2009.05.021.
3. Extended-Release Naltrexone to Prevent Opioid Relapse in Criminal Justice Offenders. Lee JD, Friedmann PD, Kinlock TW, et al. New England Journal of Medicine. 2016;374(13):1232–1242. doi:10.1056/NEJMoa1505409.
4. Methadone Maintenance vs 180-Day Psychosocially Enriched Detoxification for Treatment of Opioid Dependence. Sees KL, Delucchi KL, Masson C, et al. JAMA. 2000;283(10):1303–1310. doi:10.1001/jama.283.10.1303.
5. Prevalence and Charges of Opioid-Related Visits to US Emergency Departments. Langabeer JR, Stotts AL, Bobrow BJ, et al. Drug Alcohol Depend. 2021;221:108568. PMID:33578297. doi:10.1016/j.drugalcdep.2021.108568.
6. National Hospital Ambulatory Medical Care Survey: 2019 Emergency Department Summary Tables. Cairns C, Kang K. doi:10.15620/cdc:115748.
7. Emergency Department-Initiated Buprenorphine for Opioid Dependence with Continuation in Primary Care: Outcomes During and After Intervention. D’Onofrio G, Chawarski MC, O’Connor PG, et al. J Gen Intern Med. 2017;32(6):660–666. PMID:28194688. doi:10.1007/s11606-017-3993-2.
8. Naloxone and Buprenorphine Prescribing Following US Emergency Department Visits for Suspected Opioid Overdose: August 2019 to April 2021. Chua KP, Dahlem CHY, Nguyen TD, et al. Ann Emerg Med. 2022 Mar;79(3)225–236. PMID:34802772. doi:10.1016/j.annemergmed.2021.10.005.
9. Key Facts about Uninsured Adults with Opioid Use Disorder. Orgera K, Tolbert J. KFF. 2019 Jul 15. https://www.kff.org/uninsured/issue-brief/key-facts-about-uninsured-adults-with-opioid-use-disorder.
10. T-MSIS Substance Use Disorder (SUD) Data Book: Treatment of SUD in Medicaid, 2018. Azar A. U.S. Department of Health and Human Services. 2021 Jan 19. https://www.medicaid.gov/medicaid/data-systems/downloads/2018-sud-data-book.pdf.
11. Mobile Telemedicine for Buprenorphine Treatment in Rural Populations with Opioid Use Disorder. Weintraub E, Seneviratne C, Anane J, et al. JAMA Netw Open. 2021;4(8):e2118487. doi:10.1001/jamanetworkopen.2021.18487.
12. The Association Between Scope of Practice Regulations and Nurse Practitioner Prescribing of Buprenorphine After the 2016 Opioid Bill. Nguyen T, Muench U, Andraka-Christou B, et al. Med Care Res Rev. 2021 Apr 1. PMID:33792414. doi:10.1177/10775587211004311.
13. 2024 Mandatory Core Set of Behavioral Health Measures for Medicaid and CHIP (Behavioral Health Core Set). Medicaid. Accessed August 30, 2023. https://www.medicaid.gov/sites/default/files/2023-08/2024-bh-core-set.pdf.
14. Bridge Navigator Program. CA Bridge. Accessed May 9, 2022. https://bridgetotreatment.org/addiction-treatment/ca-bridge/bridge-navigator.
15. Buprenorphine Treatment For Opioid Use Disorder: Comparison Of Insurance Restrictions, 2017–2021. Andraka-Christou B, Simon KI, Bradford WD, et al. Health Affairs. 2023;42(5). doi:10.1377/hlthaff.2022.01513.
Contributors
Thuy Nguyen, PhD, MPA1,2; Kao-Ping Chua, MD, PhD1,2,3; Yang “Amy” Jiao, MPP1; Stephanie Lee4; Pooja Lagisetty, MD, MSc2,4,5; Amy Bohnert, PhD, MHS2,5,6; Keith Kocher, MD2,7
Affiliations: 1 Department of Health Management and Policy, School of Public Health, University of Michigan; 2 Institute for Healthcare Policy and Innovation, University of Michigan; 3 Susan B. Meister Child Health Evaluation and Research (CHEAR) Center, Medical School, University of Michigan; 4 Division of General Medicine, Medical School, University of Michigan; 5 VA Ann Arbor Healthcare System; 6 Department of Anesthesiology, Medical School, University of Michigan; 7 Department of Emergency Medicine, Medical School, University of Michigan.
Citation
Nguyen T, Chua K-P, Jiao Y, Lee S, Lagisetty P, Bohnert A, Kocher K. IHPI Brief: Use of Medications for Opioid Use Disorder After Overdoses Among Medicaid Enrollees. University of Michigan Institute for Healthcare Policy and Innovation. September 2023. Available at: https://dx.doi.org/10.7302/8133
Acknowledgments
This policy brief was supported by the IHPI Policy Sprint program, which provides funding and staff assistance to IHPI member-led teams in undertaking rapid analyses to address important health policy questions and develop products that inform decision-making at the local, state, or national level.
For more information
Please contact Eileen Kostanecki, IHPI’s Director of Policy Engagement & External Relations, at [email protected].


